CAS in Biomedical Entrepreneurship
School for Translational Medicine and Biomedical Entrepreneurship (sitem-insel School)
The CAS in Biomedical Entrepreneurship empowers professionals in life sciences, healthcare, pharma, biotech, and medtech to transform innovative ideas into market-ready ventures. Designed for both entrepreneurs and intrapreneurs, the program guides participants step-by-step from concept to commercialization, combining biomedical innovation, healthcare systems, management, financial strategy, and venture creation into one hands-on learning journey. Whether refining an existing project or developing a new idea, participants work intensively with expert coaching and mentorship to build viable products, scalable business models, and investment-ready healthcare solutions.
📞 Book a call with our program team to learn more about the CAS and how it can help advance your entrepreneurial career.
Want to Bring your Innovation to the Patient?
| Degree | Certificate of Advanced Studies in Biomedical Entrepreneurship, Universität Bern (CAS BE Unibe) |
|---|---|
| Start | 04/2027 |
| Length | April 2027 - March 2028 |
| Scope | 15 ECTS |
| Cycle | Annual |
| Flexible entry possible | No |
| Single module visitable | Yes |
| Place | sitem-insel, Freiburgstrasse 3, 3010 Bern, Switzerland |
| Language | English |
| Admission | Min. Bachelor's or higher degree in life science, biology, medicine, engineering science or equivalent. “Sur dossier” admissions possible. |
| Registration until | 15.12.2026 |
| Cost | CHF 12'600 |
| Organising institutions | School for Translational Medicine and Biomedical Entrepreneurship (sitem-insel School) |

|
About the program
CAS in Biomedical Entrepreneurship
The CAS in Biomedical Entrepreneurship is a 12-month, part-time program designed to help professionals develop, advance, and commercialize innovative healthcare projects. It focuses on the entrepreneurial process, guiding participants from the early project idea through business planning, venture creation, and financial strategy, all while addressing the realities of healthcare systems both in Switzerland and globally.
The program combines hands-on exercises, expert mentorship, and intensive coaching, enabling participants to apply the acquired knowledge directly to their corporate innovation or startup projects. Participants conclude the CAS with a market-ready strategy and a solid business plan, suitable for internal decision-making, portfolio prioritization, and implementation within established organizations or as a startup.
What You will Gain
- Take an existing project or a new idea and develop it towards market readiness
- Acquire practical skills in business strategy, venture creation, and financial planning
- Gain insights into Swiss and global healthcare systems
- Benefit from individual coaching and mentorship throughout the program
- Connect and exchange knowledge with peers, lecturers, and industry experts
- Receive an internationally recognized certificate from the University of Bern
Who Should Apply?
This program is designed for:
- Entrepreneurs and intrapreneurs in life sciences, healthcare, pharma, biotech, and medtech aiming to advance innovative projects or statups
- Professionals from industry, hospitals, or academia seeking to gain practical skills and entrepreneurial knowledge to translate ideas into viable healthcare solutions
To ensure confidentiality and protect sensitive corporate and project-related information, all coaches, lecturers, and participants are required to sign a non-disclosure agreement (NDA). Participants and/or their employing organizations retain full ownership of all ideas, data, and intellectual property at all times. The program supports innovation development without any transfer of rights or claims by the University or involved experts.
Program structure
Program Structure
The CAS in Biomedical Entrepreneurship is a 12-month, part-time program worth 15 ETCS credits where participants work hands-on on their own project. The program covers the following key topics:
- Biomedical Innovation
- Healthcare Systems & Market Context
- Leadership & Management
- Financial Strategy
- New Venture Creation
Throughout the CAS, participants continuously develop their own entrepreneurial project with structured coaching, culminating in a final business plan. Class sizes are small, typically up to 15 participants, ensuring personalized support and interactive learning.
On-site dates are very limited to accommodate the schedules of working professionals. The majority of the program is delivered online in a flexible blended learning format, combining self-paced study with interactive online sessions.
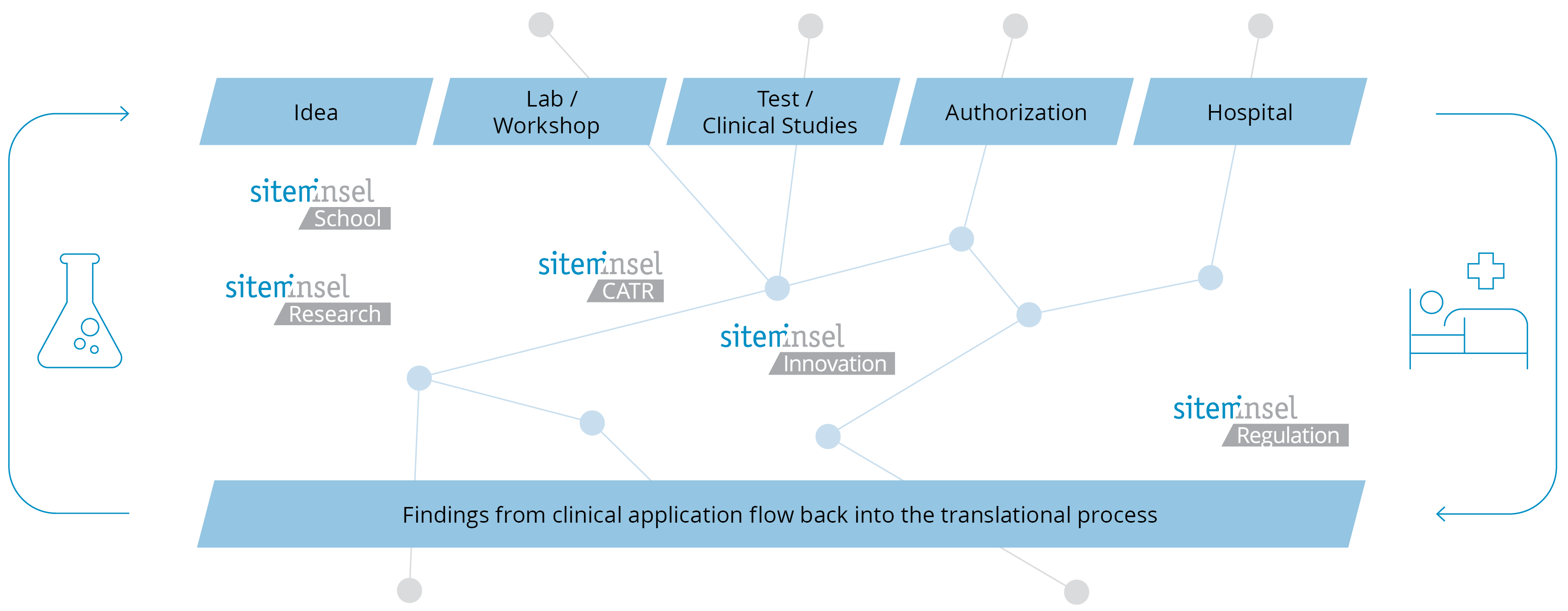
The CAS Biomedical Entrepreneurship is part of a modular program in Translational Medicine and Biomedical Entrepreneurship. Participants can seamlessly continue with the CAS in Translational Medicine if desired. Completing these two CAS programs, together with two additional modules and a more extensive MAS project, leads to the MAS in Translational Medicine and Biomedical Entrepreneurship (MAS TMBE).
Module content
Biomedical Entrepreneurship
From Idea to Market: Turning Innovation into Impact
Biomedical entrepreneurship focuses on creating, developing, and managing new ventures to bring innovative healthcare products and services to the market. Module 6 equips participants with the essential entrepreneurial skills to combine scientific knowledge with business strategies, covering creativity, innovation, business strategy, product management, negotiation, and leading entrepreneurial teams. Throughout the program, participants work on their own innovation project, receiving individualized coaching to develop it into a viable venture and culminating in a final business plan that demonstrates market readiness. This hands-on approach prepares professionals to launch and lead successful biomedical ventures while strengthening their career in life sciences, healthcare, pharma, biotech, and medtech.
The module is integral part of the MAS in Translational Medicine and Biomedical Entrepreneurship (TMBE) program or is offered as a shorter standalone CAS in Biomedical Entrepreneurship.
Learning objectives
Upon completion of this module, participants should be able to:
- Demonstrate an understanding of economic and entrepreneurial principles by analyzing key factors influencing the development and commercialization of therapeutic products
- Apply entrepreneurial thinking to science-based innovation by integrating business strategies, market analysis, and commercialization pathways
- Evaluate stakeholder dynamics and project management strategies by identifying key industry players, business management concepts, and approaches relevant to life sciences
- Develop and assess business models for therapeutic products by translating ideas into structured plans, evaluating market potential, and understanding foundational aspects of enterprise creation and growth
Schedule
This module runs over one year and includes few on-site days in Bern (approximately 1-2 events per month) combined with intensive individual business coaching to advance each participant’s project or startup. The program is further complemented by blended learning, online activities, and self-paced assignments. The on-site days are the only in-person requirement.
The on-site dates for spring 2027 will be anounced soon. Contact the school if you have any questions about course dates.
Across the program, an average attendance of 80% is expected, so participants can occasionally miss a session without affecting completion. Whenever possible, virtual participation is offered to increase flexibility for working professionals or international participants.
Module leaders & lecturers
Director of the Department of Management and Entrepreneurship, University of Bern

Prof. Dr. Artur Baldauf
Artur Baldauf is Professor of Business Administration at the University of Bern, specializing in Strategic Business Management. After his doctorate at the Vienna University of Economics and Business Administration and his habilitation at the University of Vienna, he has taught and researched at various European and US universities. Many projects have been implemented in cooperation with renowned, international companies.
Organising institution and faculty
Swiss Institute for Translational and Entrepreneurial Medicine (sitem-insel)
sitem-insel is the Swiss Institute for Translational and Entrepreneurial Medicine. It is located on the Insel Campus Bern and benefits from its proximity to Switzerland's largest university hospital (Inselspital) as well as the University of Bern. At sitem-insel, a wide variety of units from clinics, industry, research, and education are networked under one roof, driving innovation for the benefit of patients. Our mission is to establish, operate and develop a National Center of Excellence for Translational Medicine that professionalizes translational research for the benefit of patients, society, and science. An onsite cutting-edge 20,000 m2 facility is the sitem-insel catalyst for a multidisciplinary collaborative approach to unlocking ‘bench to bedside’ innovation.
Bringing Innovation to the Patient – by Connecting People.
How to get a research idea from bench to bedside?
Together with you, we are committed to bridging the gap between clinical practice, research, entrepreneurship and regulation.
Medical Faculty of the University of Bern
Our education programs in translational medicine, biomedical entrepreneurship, regulatory affairs and artificial intelligence are offered under the umbrella of the Medical Faculty of the University of Bern.
The Medical Faculty is known for an excellent curriculum in human and dental medicine, and also offers an attractive and high-quality range of training and continuing education in the field of health and medicine. With several institutes and clinics in various disciplines, it has excellent, internationally recognized research performance. It also conducts cutting-edge research in artificial intelligence for medicine and provides access to data science for all research groups.
Experts from industry and academia
Our experts from academia, clinic, industry and regulatory authorities, carry the teaching responsibility and support us in educating healthcare and regulatory specialists, bridging knowledge gaps, and advancing professional careers. They combine deep theoretical knowledge from their innovative research, with the latest practical insights from their extensive work with industry.
Admission, fees and installments
Admission requirements
Applicants must hold at least a Bachelor's degree from a university or university of applied sciences in the fields of natural sciences and engineering, medicine, pharmacy or law. Professional experience is not mandatory. If the applicant has no prior academic degree or professional experience, the study commission may define further conditions for the applicant to successfully complete the course.
Admission process
Please submit your complete application including all required attachments via the registration form. Extensions of application deadlines may be granted by the Directorate.
To complete the application, you will need the following documents:
- Copy of your resume/curriculum vitae (CV)
- Recent passport photo (see guideline) and a copy of the passport/ID
- Signed copy of Confidentiality Declaration (see documents below)
- Copy of highest relevant academic degree
Once we have received your application, we will gladly evaluate your documents and get back to you within a few working days. Upon successful registration, you will get a Campus Account from the University of Bern, which allows access to the student area of the University websites, the University´s WLAN network (eduroam), the use of library databases and of e-journals. MAS students will also receive a UNICARD and have access to sports, childcare and counselling facilities offered by the University of Bern.
Deadlines
Application for scholarship: May 31
Application for fall term: July 15
Application for spring term: December 15
Standalone modules can be admitted anytime, provided places are still available.
The Directorate may grant exceptions on the above deadlines.
Tuition fees & installments
Master of Advanced Studies (MAS): CHF 31'500.–
Diploma of Advanced Studies (DAS): CHF 23'100.–
Certificate of Advanced Studies (CAS): CHF 12'600.–
The full study fee can be paid in full at once or in installments per semester.
There is no entitlement to a refund or waiver of the course fees if parts of the course are not attended. Costs for travel, accommodation and catering are not included. Insurance is in the sole responsibility of participants (accident, travel, cancellation, etc.).
Withdrawal of the registration before the registration deadline is possible without cost consequences. In case of withdrawal after the registration deadline, the full course fee must be paid.
Get in Touch

- Name / Titel
- Robin Bétrisey
- Funktion
- Managing Director
- school.sitem@unibe.ch
Associate Courses
CAS in Translational Medicine
| Degree | CAS |
|---|---|
| Start | 09/2026 |
| Language | Englisch |
| Cost | CHF 12'600 |
The part-time CAS in Translational Medicine at University of Bern equips professionals to transform biomedical research into clinical and commercial healthcare solutions. Gain practical skills in R&D strategy, intellectual property, and clinical trial design while connecting with industry experts and peers.
MAS in Translational Medicine and Biomedical Entrepreneurship
| Degree | MAS |
|---|---|
| Start | 09/2026 |
| Language | Englisch |
| Cost | CHF 31'500 |
The program includes all relevant aspects of the translational process – entrepreneurial know-how in addition to all basic scientific, clinical, and technical knowledge. It covers different points relevant for the development and commercialization of medicinal products and medical devices.
CAS in Artificial Intelligence in Medical Imaging
| Degree | CAS |
|---|---|
| Start | 02/2027 |
| Language | Englisch |
| Cost | CHF 12'600.- |
The Certificate of Advanced Studies in Artificial Intelligence in Medical Imaging (CAS AIMI) offers a hands-on, application-focused introduction to Artificial Intelligence (AI) in the field of medical imaging. Designed specifically for professionals without relevant technical background, it provides the foundational understanding and practical skills needed to critically assess, use, and contribute to AI solutions in clinical and research environments. Through real-world case studies and guided exercises, participants will learn how AI is transforming diagnostic imaging and healthcare delivery.
CAS in Regulatory Affairs
| Degree | CAS |
|---|---|
| Start | 09/2026 |
| Language | Englisch |
| Cost | CHF 12'600 |
The part-time CAS program in Regulatory Affairs provides practice-oriented training in the European regulatory framework for medical devices and in vitro diagnostic (IVD) devices, including the latest developments of the EU Medical Device Regulation (MDR). Participants gain practical expertise in regulatory strategy, product classification, compliance, and lifecycle management while building a strong professional network with industry experts and peers.